 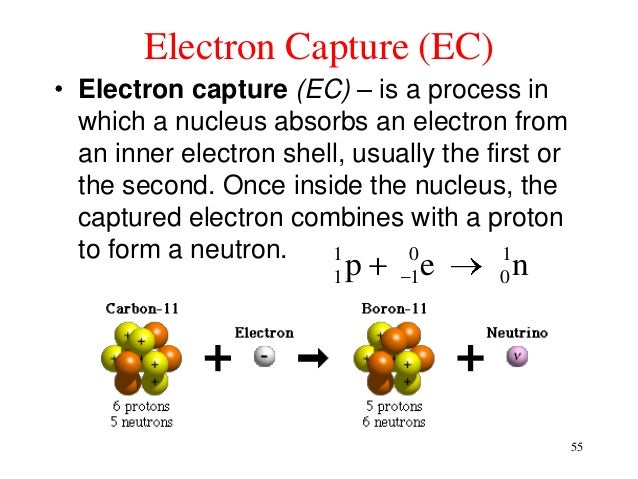
Presented here are the correlation of protonated sites to types of product ions unique in ECD and the importance of unoccupied MO's to understand the bond cleavage reactions connected with the Franck-Condon process. This process will reduce the atomic number by one and not changed the atom's mass. The result is that a proton will combine with this electron and a neutron is formed. Positrons have the same mass as an electron but are positively charged. Thus, the atomic number decreases by 1, but the. That is, the electron is captured by the nucleus, which loses a proton and gains a neutron (which is about the same mass as the proton). A typical beta decay process involves carbon-14, often used in radioactive dating techniques. Electron capture is when the nucleus takes in an electron, combines it with a proton (due to their opposite charge), and thus forms a neutron. As is the case with -emissions, -emissions are often accompanied by -radiation. In the process, the atomic number increases by one while the atomic weight stays the same.  
Based on the calculated energy and the molecular geometry for various stages of species concerned with the ECD process, mechanisms for proton transfer and electron capture through the Franck-Condon process were discussed. Electron capture occurs when an inner-orbital electron (negatively charged) is captured by the nucleus (positively charged). The electron is then ejected from the nucleus. We carried out MO calculations on the ECD processes of glycylserine as a model peptide to elucidate how the process of proton transfer correlates with backbone cleavages. This proton transfer produces various types of protonated peptides that should be responsible for the cleavage after electron capture. Application of electron-capture dissociation (ECD) to the tetramer causes unfolding starting from the C-terminal domain, in good agreement with solution denaturation data, and. 2 Analytical Research and Development, Pfizer Inc., Chesterfield, MO, 63017, USA. Against a mechanism presented so far that amide bonds catch the hydrogen atom released from the protonated peptide through electron capture, we present another mechanism focusing the proton rearrangement among the sites of the nitrogen and the oxygen atoms. Electron capture in an electronmolecule reaction is enabled by excited states of the corresponding anion. 1 Department of Chemistry, Washington University, St. Electron capture is a process in which the proton-rich nucleus of an electrically neutral atom absorbs an inner atomic electron, usually from the K or L. A novel approach was proposed for elucidating the reaction mechanism of the electron capture dissociation (ECD) recently introduced powerful technique to characterize peptides and proteins. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
